Thus all isotopes have a same atomic number. But in the case of isotopes, all the isotopes have a same number of electrons. No, each element possesses an individual atomic number, acting as a unique identifier. Can two Elements share the Same Atomic Number? That is why in the above table of an element’s mass and atomic number the mass number is more than the atomic number 3. 
The primary difference between them is that atomic number is the number the protons in an atom while mass number is the sum of protons and neutrons. What are Some Differences between Atomic Mass and Atomic Number? The position of an atom in the periodic table is determined by its atomic number. What is Atomic Mass and Atomic Number?Ītomic mass as we are getting from the name, is the total mass of an atom and is given by the total number of protons and neutrons we are not counting electrons because their mass is negligible.Ītomic number is equal to the number of protons or the number of electrons in any element. 
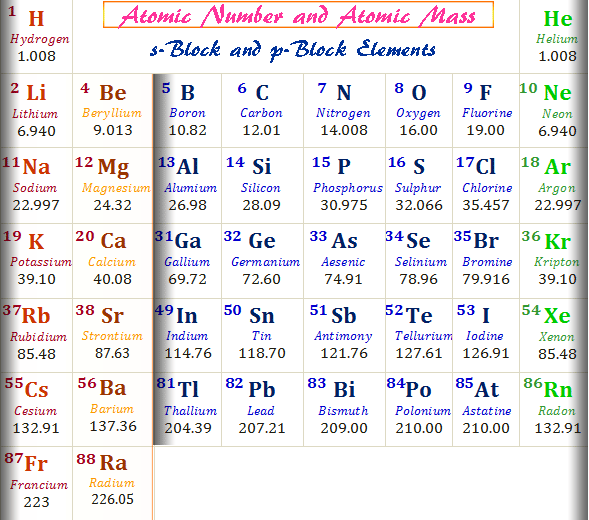
The following table provides the information about Atomic Mass and Number of first 20 elements.Ītomic Mass and Atomic Number – FAQs 1. The atomic number is the whole number typically placed above or within the element’s symbol on the periodic table.Įlements Table with Atomic Mass and Atomic Number It is usually not a whole number because it includes the masses of protons, neutrons, and electrons, each with fractional masses.Īlways a whole number since it represents the count of protons in an atom’s nucleus.Ītomic mass varies from element to element and even between isotopes of the same element.Ītomic number is unique to each element and defines its identity on the periodic table.Ĭarbon-12 has an atomic mass of approximately 12.01 u due to its mixture of isotopes.Ĭarbon-12 has an atomic number of 6 because it has 6 protons.Ītomic mass is useful for calculating the mass of a sample of atoms or molecules and for determining molar mass.Ītomic number is crucial for identifying elements and organizing them in the periodic table.Ītomic mass is typically measured in atomic mass units (u) or grams per mole (g/mol).Ītomic number is dimensionless it’s just a whole number count.įound on the periodic table beneath the element’s symbol or as a decimal value in the element’s box. The number of protons in the nucleus of an atom. The total mass of an atom, including protons, neutrons, and electrons. There are various differences between atomic mass and atomic number, some of those difference between Atomic and Mass Number are listed in the following table: From that, we have selected some examples for atomic mass, which are provided in the following table. There are 118 elements known in the modern-day periodic table. Thus, for oxygen-16 (16O), the atomic mass (mass number) is 16. So, for carbon-12 (12C), the atomic mass (mass number) is 12.Ītomic Number (Z) = 8 (since oxygen has 8 protons) Role of Mahatma Gandhi in Freedom Struggle. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
